Capillary Action
Over View
After watching the above video, you may wonder why coffee slowly creeps up in the paper towel, that too against gravity. What phenomenon is responsible for this? The short answer is it is the capillary action. Other names for this phenomenon are capillary motion or wicking.
Let us understand what causes this interesting phenomenon.
Cohesion Vs Adhesion
Before looking into what causes capillary action, we need to understand the concept of adhesive and cohesive forces in liquids.
Consider a glass of water. We know that it consists of billions and billions of water (H2O) molecules. Each molecule experiences strong intermolecular attraction forces, known as Van de Waals force, from the neighboring molecules because of electrostatic bonding.
When these intermolecular attraction forces are between like molecules, say water molecules, they are referred to as cohesive forces.
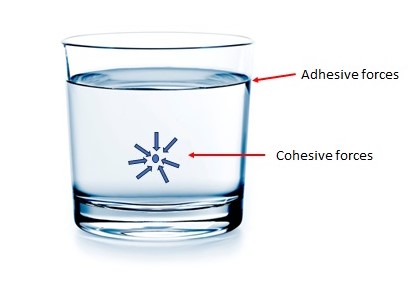
If these intermolecular attraction forces are between unlike molecules, say between water molecules and glass molecules, then we call them adhesive forces.
Now let us see how the combination of these adhesive and cohesive forces in a liquid result in capillary motion in a capillary tube.
Capillary Action
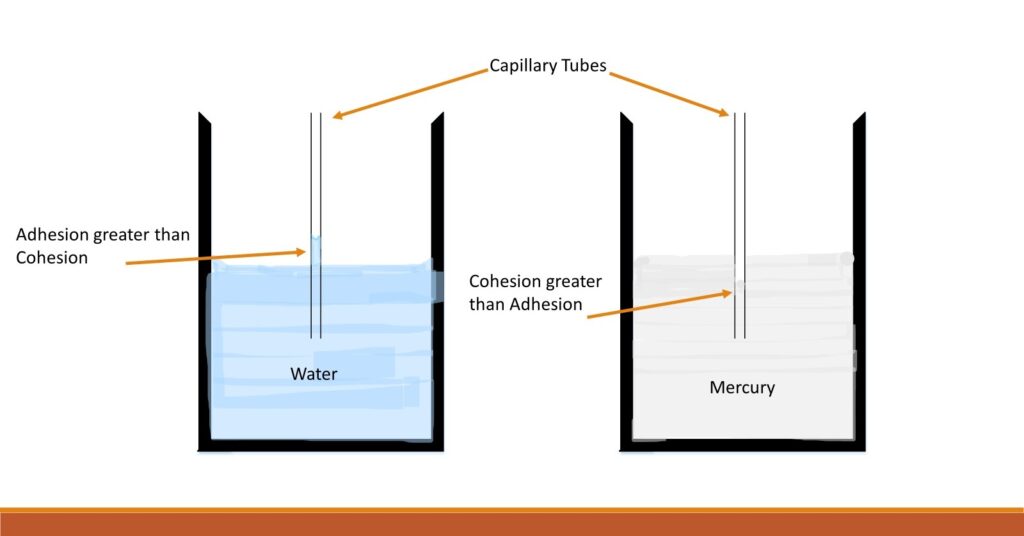
Compare the two capillary tubes in the picture above. Notice that in the capillary tube in water, water has raised above the level in the container. This happens because the adhesion between water and glass of the capillary tube is greater than the cohesion between water molecules. So, water molecules slowly creep up in the capillary tube.
How high water rises in the capillary tube depends on several factors such as the radius of the capillary tube, the density of the fluid, and the temperature. The smaller the radius of the capillary tube, greater the height reached. And lower the density of the fluid, higher the height reached.
However, in the case of mercury, what happens inside the capillary tube is more interesting. It goes below the level in the container. This happens because the cohesion in mercury is greater than the adhesion between mercury and glass. So, mercury creeps down in the capillary tube.
In this context, it is to be noted that cohesion in fluid is responsible for another interesting phenomenon: Surface Tension.
In the next section, let us explore some of the applications we commonly encounter on daily basis.
Applications

Burning Candles
What makes a candle burn steadily? It is the capillary motion.
The hot liquid wax slowly moves up the wick, through the narrow capillaries of the wick, to get converted into vapor which burns at the top of the wick.

Paper Towels &
Microfiber Cloths
When we spill something on the kitchen counter, our best friend that helps us in the cleanup process is a piece of paper towel. It is so amazing that it quickly absorbs most of the fluid in seconds. How does it do that?
It is the same capillary motion that does the trick. Paper towel is very porous with thin capillaries. As we place a piece of paper towel on the spilled fluid, it quickly rises from the counter to the paper towel through the capillaries. Thereby helping us in the cleanup process.

Bath Towels
Just like paper towels, bath towels that we use after shower or bath work in the same way. The porous material in the towels help quick absorption thanks to wicking again.

From Roots To The Top
Of Trees
Have you ever wondered how water flows to different parts of a tall tree, especially to the top defying gravity? Again, this happens, in most of the trees, thanks to capillary motion.
However, in very tall trees, like the red wood trees, it is not only capillary action that helps to transport water from the ground to the top, but also other factors like the pressure gradient created by the evaporation of water on the surface of leaves.
References
- https://www.usgs.gov/special-topics/water-science-school/science/capillary-action-and-water#:~:text=Capillary%20action%20occurs%20when%20the,and%2C%20of%20course%2C%20gravity.
- https://www.thoughtco.com/definition-of-capillary-action-604866 (Helmenstine, Anne Marie, Ph.D. “Capillary Action: Definition and Examples.” ThoughtCo, Aug. 26, 2020, thoughtco.com/definition-of-capillary-action-604866.)
- https://pressbooks.uiowa.edu/clonedbook/chapter/cohesion-and-adhesion-in-liquids-surface-tension-and-capillary-action/
