Niels Bohr
Pioneer Of
Quantum Physics

Introduction
Niels Bohr, a Danish theoretical physicist and a pioneer of quantum physics, stands as one of the most influential figures in the history of physics. Renowned for his groundbreaking work in atomic structure and quantum mechanics, Bohr’s contributions not only reshaped scientific understanding but also laid the foundation for much of modern physics.
He was awarded the Nobel prize in 1922 in physics for his work “on the model of the atom and the consequence of light emanating as electrons jump from one energy state to another”.
This article explores his key discoveries, his enduring legacy, and the profound impact he had on quantum physics.
State of atomic theory
Before Bohr
Prior to Bohr, atomic models like those proposed by Thomson and Rutherford relied on classical physics. However, these theories were unable to account for several important atomic phenomena:
- Understanding how atoms maintain their stability was a major problem that classical physics could not solve.
- The reason each element produces its own unique spectral lines remained unclear.
- Classical physics did not provide a satisfactory explanation for how electrons behave within an atom.
Thomson Atom Model

J. J. Thomson, who discovered the electron in 1897, introduced an atomic model in 1904. He suggested that atoms are made up of a uniform sphere of positive charge, with electrons scattered throughout—much like plums in a pudding as the picture below illustrates.
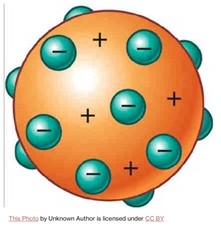
This became known as the ‘plum-pudding’ model.
Rutherford Atom Model

Ernest Rutherford at the University of Manchester, England, after conducting an experiment involving alpha particles striking a gold foil, discovered the presence of a positively-charged-tiny-heavy nucleus in an atom.
According to Rutherford’s model, an atom consists of a tiny, dense, central nucleus surrounded by orbiting negatively charged light electrons as the picture below shows.
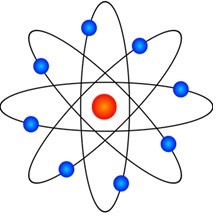
The problem with this model is: In an atom, if electrons revolve around the nucleus, like the planets around the sun, they should radiate energy, according to classical electrodynamics, and collapse into the nucleus. Thereby, making an atom unstable. Evidently, that is not the case.
Therefore, Rutherford model could not explain the stability of an atom. In addition, it could not account for the characteristic spectral lines produced by the gaseous state of different elements when they are excited.
Bohr Atom Model
While working under the leadership of Rutherford at the University of Manchester and expanding upon Rutherford’s earlier atomic model, Bohr introduced a new atomic model in 1913. This development is widely regarded as a significant scientific achievement. In recognition of his contribution, Bohr was awarded the Nobel Prize in 1922.
Inspired by the emerging quantum theory, proposed by Max Planck and Albert Einstein, Bohr introduced this new principle to address the inconsistencies in the earlier atomic models related to electron behavior in an atom.
Key Features:
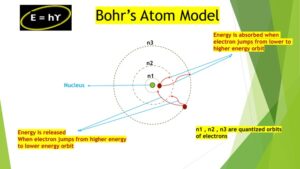
Bohr proposed that:
- Electrons in an atom orbit the central heavy nucleus in discrete energy levels, or “shells” as the picture above illustrates.
- Electrons can only exist in discrete allowed orbits with specific energy values. These orbits are called quantized orbits.
- While the electrons stay in a particular quantized orbit, they do not emit energy.
- These quantized-orbits prevent electrons spiraling into the nucleus, ensuring the stability of an atom.
- Atoms emit or absorb energy only when electrons jump between these different quantized orbits.
These concepts not only elegantly explained the stability of an atom, but also why atoms emit light at specific wavelengths. Bohr theoretically predicted the wavelengths for hydrogen. His prediction matched the observed spectral lines of hydrogen.
Bohr’s model incorporated early quantum concepts and successfully resolved inconsistencies between classical physics and experimental results. His ideas also introduced the principle of quantization to atomic systems, a notion that would prove foundational for the development of quantum mechanics.
Limitation of Bohr model
Bohr’s model was effective mainly for hydrogen-like atoms. It could not explain more complex atoms. Nor could it explain the fine spectral lines. These limitations led to the development of more advanced quantum theories such as wave mechanics by Schrodinger.
Bohr’s Legacy
Niels Bohr’s legacy extends far beyond his scientific papers. As a mentor and leader, he established the Institute for Theoretical Physics in Copenhagen, which became a global center for research and collaboration. His institute attracted and nurtured some of the most brilliant minds of the twentieth century, including Werner Heisenberg, Wolfgang Pauli, and many other future Nobel laureates.
Bohr’s philosophical approach to science, especially his advocacy of the complementarity principle, influenced the interpretation of quantum mechanics. This principle held that certain properties (like wave and particle aspects of light) are complementary and cannot be observed simultaneously, shaping the way physicists understand the dual nature of matter and energy.
In addition to his scientific work, Bohr was dedicated to promoting open scientific dialogue and international cooperation. After World War II, he became an advocate for the peaceful use of atomic energy and transparency among nations, supporting efforts that eventually led to the establishment of CERN and the Atoms for Peace initiative.
Pioneer of Quantum Physics
Bohr’s work is inseparable from the birth and evolution of quantum physics. By introducing quantum concepts into atomic theory, he provided the first successful explanation for atomic stability and spectral lines. His interactions with contemporaries such as Einstein, Schrödinger, and Heisenberg fueled debates that clarified the philosophical and theoretical foundations of quantum mechanics.

Starting in 1927 at a Solvay conference and spanning two decades, Bohr and Einstein engaged in a cordial yet rigorous debate concerning the interpretation of quantum physics. Einstein consistently challenged Bohr’s position that the microworld of atoms and molecules is fundamentally indeterminate, seeking to refute Bohr’s arguments. However, Bohr continually provided substantive responses to Einstein’s criticisms, and Bohr’s reasoning ultimately prevailed.
The Copenhagen interpretation of quantum mechanics, to which Bohr was central, remains one of the most widely taught frameworks for understanding quantum phenomena. Bohr’s insistence on the necessity of probabilistic and statistical interpretations in quantum physics marked a decisive break from deterministic classical physics, forever changing the course of scientific thought.
His insights continue to inspire research in quantum information, particle physics, and cosmology. Concepts rooted in Bohr’s work underpin technologies such as lasers, semiconductors, and even emerging fields like quantum computing.
Conclusion
Niels Bohr’s discoveries, legacy, and influence on quantum physics are profound and far-reaching. His pioneering work not only explained the mysteries of the atom but also revolutionized the way scientists view the universe. Through his scientific achievements, mentorship, and commitment to the scientific community, Bohr’s impact endures, shaping the trajectory of physics and technology for generations to come.
References
- Quantum: Einstein, Bohr, and the Great Debate About the Nature of Reality by Manjit Kumar
- Great Physicists: The Life and Times of Leading Physicists from Galileo to Hawking, by William H. Cropper
- Aaserud, Finn. “Niels Bohr”. Encyclopedia Britannica, 14 Nov. 2025, https://www.britannica.com/biography/Niels-Bohr. Accessed 12 December 2025.
- Britannica Editors. “Rutherford model”. Encyclopedia Britannica, 7 Nov. 2025, https://www.britannica.com/science/Rutherford-model. Accessed 18 December 2025.
- Stewart, Ken. “atomic model”. Encyclopedia Britannica, 27 Nov. 2025, https://www.britannica.com/science/atomic-model. Accessed 22 December 2025.
